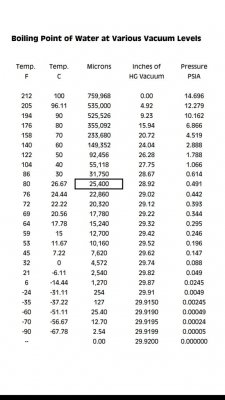
I don't know. The chart I looked up shows it boiling down to -90 so if it's boiling it must still be a liquid.
Pressure has lots of eye defects on boiling and freezing points. We put coolant in our car for antifreeze and it's that ethylene glycol or whatever other chemicals it may be made out of that lowers the freezing point below 32 but the boiling point is strictly increased just because of the 14 to 16 pounds of pressure it's under.
So I assume in a vacuum situation it's possible to have water a lot colder than 32 and not be solid. I know on the slushie machines and Ice chip machines with the slow constant agitation and the ice crystals the temperature can be well below 32 but not solid with no chemicals to depress the freezing point.
There's actually some science that goes in to those freeze machines.
Pressure has lots of eye defects on boiling and freezing points. We put coolant in our car for antifreeze and it's that ethylene glycol or whatever other chemicals it may be made out of that lowers the freezing point below 32 but the boiling point is strictly increased just because of the 14 to 16 pounds of pressure it's under.
So I assume in a vacuum situation it's possible to have water a lot colder than 32 and not be solid. I know on the slushie machines and Ice chip machines with the slow constant agitation and the ice crystals the temperature can be well below 32 but not solid with no chemicals to depress the freezing point.
There's actually some science that goes in to those freeze machines.